This happens when the bonds in a molecule are arranged in a way in which polarities cancel each other. Some molecules have polar bonds but no dipole moment. When placed in an electric field, these molecules tend to align themselves with the electric field. A polar molecule is one that shows an imbalance in its electron distribution. Knowledge of both the bond polarities and the shape are required in the determination of the molecule’s overall polarity (dipole moment). 
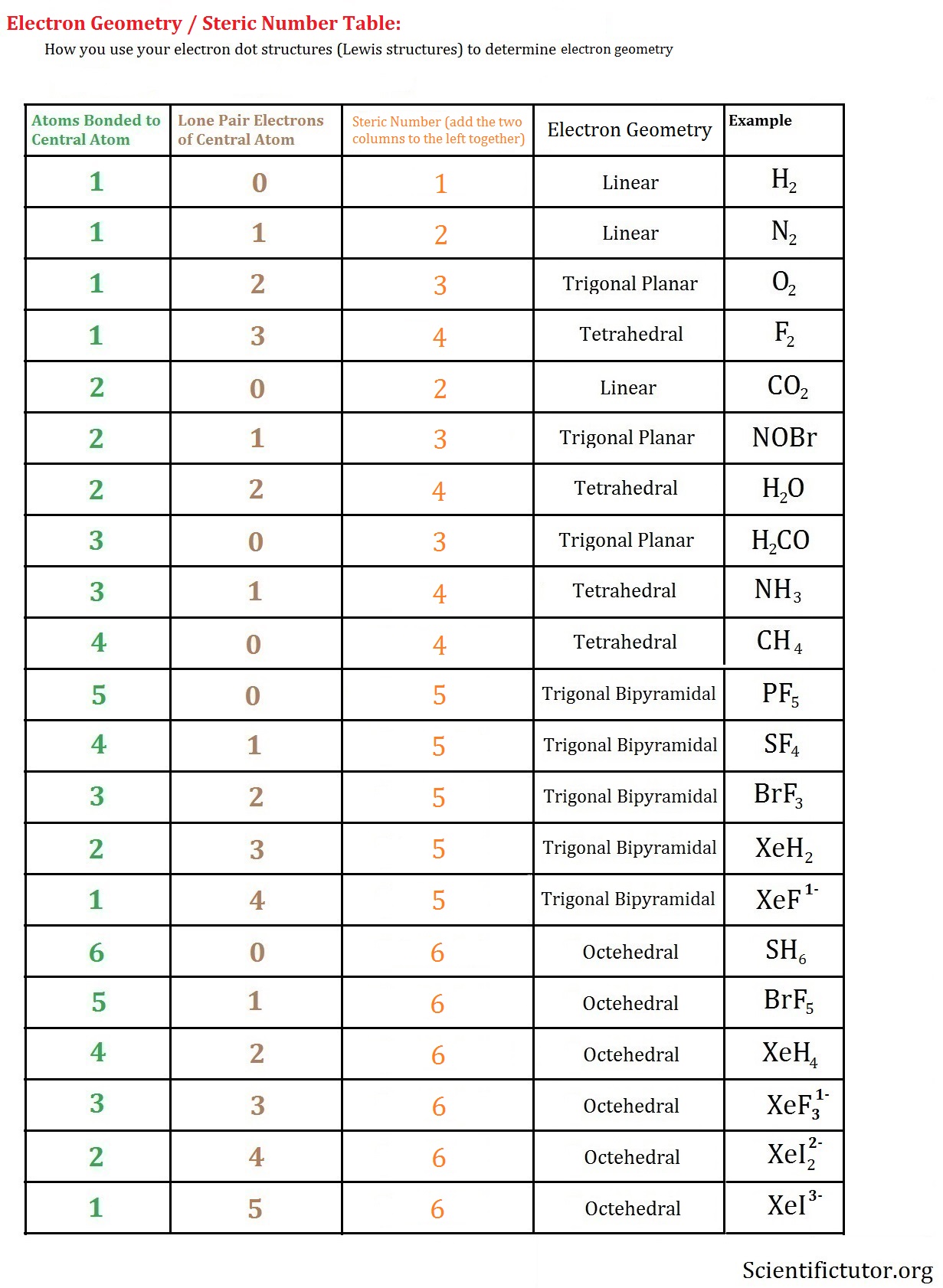
Bonds (single, double, and triple bonds count as one (1) area of electron density or one effective electron pair).īefore performing this exercise, please review (in your text or notes) the various geometries and bond angles that can be produced by different numbers of effective electron pairs around the central atom.Ī polar bond is one in which the electron cloud is closer to the nucleus of one atom (the more electronegative one) than the other (the less electronegative one).Lone pairs of electrons: these electrons tend to take more space than the bonded pairs in space leading into somewhat distorted structures.The shape of a molecule is then determined by the number of areas of electron density (or, number of effective electron pairs) around a central atom. Both the molecular geometry and the polarity of individual bonds then determine whether the molecule is polar or not.īefore determining the shape of a molecule, the Lewis structure must be properly drawn. The main postulate for the VSEPR theory is that the geometrical structure around a given atom is principally determined by minimizing the repulsion between effective electron pairs. The VSEPR (Valence Shell Electron Pair Repulsion) model is used to predict the geometry of molecules based on the number of effective electron pairs around a central atom. Note that any valid resonance structure of a molecule can be used to determine its shape and polarity. Resonance structures can also be non-equivalent, in which case they will have different numbers and/or locations of bonds. The molecule SO2 (shown above) has two such resonance forms. These structures are often equivalent, meaning that they contain the same number of bonds at different locations. Resonance refers to bonding in molecules or ions that cannot be correctly represented by a single Lewis structure. This includes rules for structures which obey the octet rule as well as those which involve expanded or reduced octets and how to calculate and optimize formal charge. 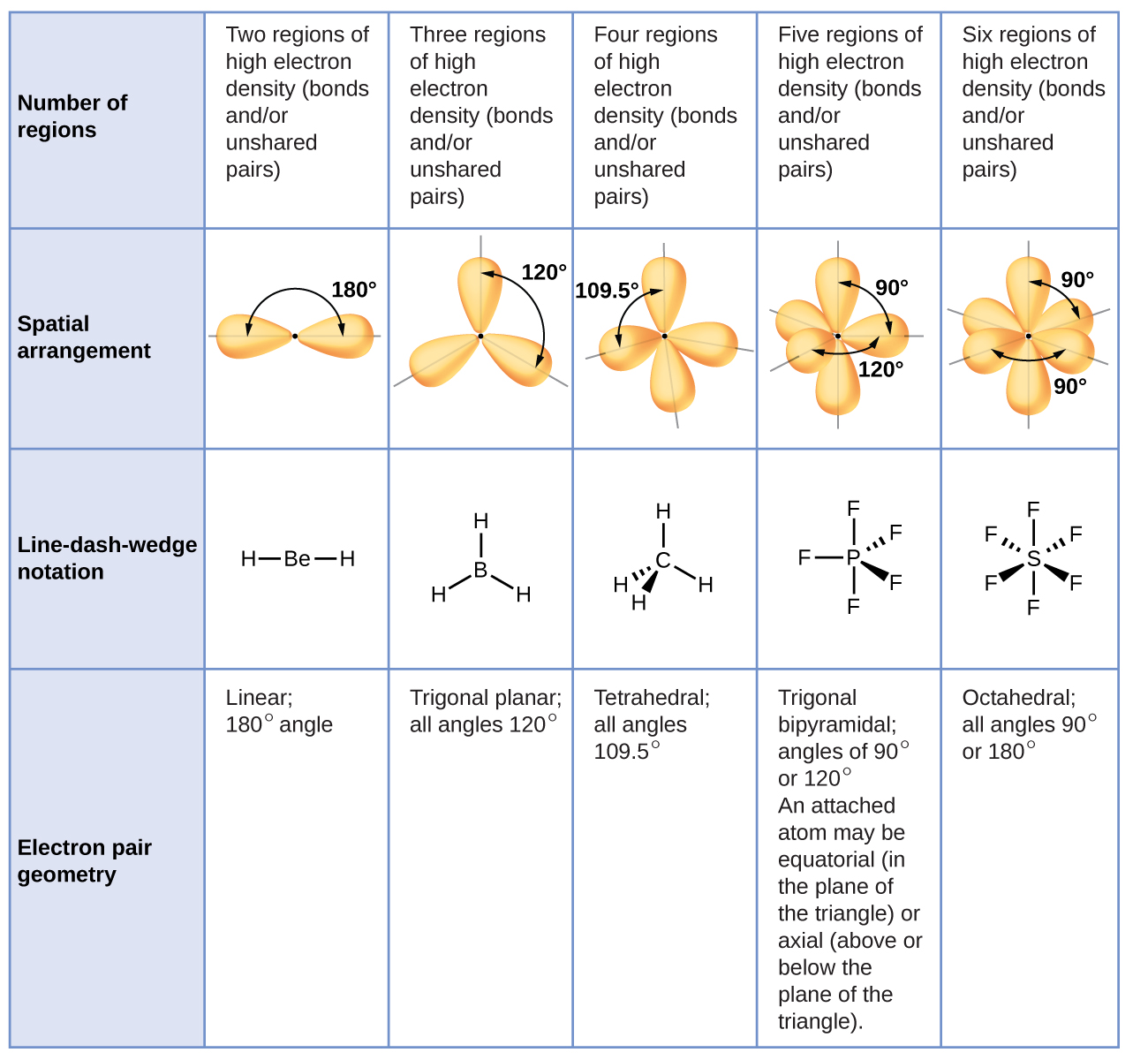
Please review (in your text or notes) the rules for drawing Lewis structures before performing this exercise. Examples are shown for the molecules SF2 and CH2O below. Pairs of dots are used to represent lone pair electrons. Atoms can even share three pairs of electrons, represented by three short lines (a triple bond). 
Sometimes atoms can share two pairs of electrons, represented by two short lines (a double bond). A shared pair of electrons is represented as a short line (a single bond). A Lewis Structure is a representation of covalent molecules (or polyatomic ions) where all the valence electrons are shown distributed about the bonded atoms as either shared electron pairs (bond pairs) or unshared electron pairs (lone pairs). Today you will be creating structures named after Lewis, as well as VSERP theory to predict bond angle and hybridization. The circumstances behind his death are unclear. Sadly, Lewis was found dead in his Berkley laboratory after working with hydrogen cyanide. The ‘greatest scientist to never win the Nobel prize ‘, Lewis was nominated for the Nobel award 41 times, but evidence exists that his nominations were blocked for political reasons stemming from early criticisms of his graduate mentor while working in Germany (Kean, 2011). Lewis mentored countless PhD students including twenty Nobel laureates, many of which collaborated with Lewis. Lewis also coined the word ‘photon’ as ‘light quanta’ in a 1926 letter to Nature. His prolific innovations span work in acid base theory, thermodynamics, covalent bond theory and the behavior of electrons in the covalent bond. It would be hard to overstate Gilbert Lewis’ contributions to modern chemistry. Return your disassembled model kit to the back shelf.Dispose of naphthalene in the labeled beaker in hood 1.Dispose of solvents in the labeled beaker in hood 1.Wear splash goggles when anyone in the lab uses solvents or chemicals.Wear splash goggles when anyone in the lab uses solvents or chemicals To determine the hybridization of the central atoms, the number and types of bonds, the geometries, and the polarities of the molecules and ions.To draw Lewis structures (both projection and perspective drawings) for each of these molecules and ions.To analyze bond angles of a variety of molecules and ions using molecular model kits.Polarity *Note: This is the polarity of the molecule, not the bond polarity Quiz on the tabled structures via BB before lab.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
